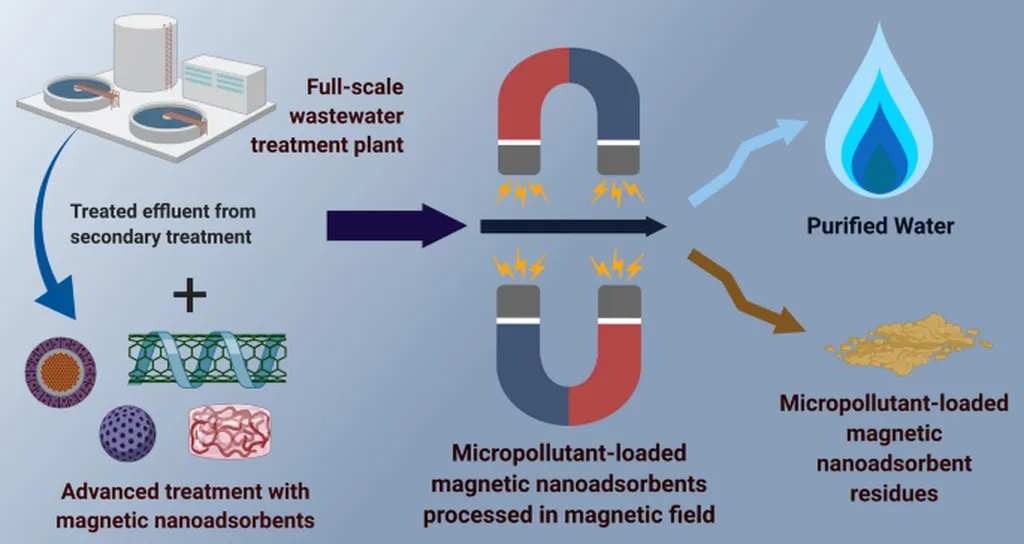
In the quiet corridors of Alborz University of Medical Sciences in Karaj, Iran, a team led by environmental health engineer Sakineh Mollai Tavani has unlocked a promising pathway to cleaner water—one that could ripple across industries, particularly energy. Their discovery? A paramagnetic polymer-based nanoadsorbent that efficiently captures two common painkillers, ibuprofen and naproxen, from water supplies. These pharmaceuticals, often found in trace amounts in wastewater, are not only persistent but increasingly scrutinized for their ecological impact.
“What we’ve developed isn’t just another adsorbent—it’s a smart one,” says Mollai Tavani. “It combines the magnetic responsiveness of iron oxide nanoparticles with the chemical selectivity of poly(4-vinylpyridine), or P4VP. This dual nature allows rapid uptake of contaminants and easy separation under a magnetic field.” The synthesis, confirmed through SEM, TEM, FTIR, and VSM analyses, yields a hybrid material with uniformly dispersed Fe₃O₄ domains and abundant pyridinic sites—functional groups that bind strongly to aromatic pharmaceutical molecules.
The implications for the energy sector, especially in water-intensive operations like oil and gas extraction, cooling systems, and desalination, are significant. Traditional filtration methods struggle with trace pharmaceuticals, often requiring costly multi-stage treatment. But Fe₃O₄–P4VP nanoparticles could streamline this process. Under optimal conditions, the team achieved 90.42% removal of ibuprofen and 92.42% of naproxen, with adsorption capacities of 18.08 mg/g and 92.7 mg/g, respectively. “These are not marginal improvements,” notes Mollai Tavani. “They represent a leap toward meeting stringent discharge limits without overhauling existing infrastructure.”
Kinetic studies revealed that adsorption follows a pseudo-second-order model, suggesting chemisorption—meaning the interaction isn’t just physical but involves chemical bonding. Meanwhile, Freundlich isotherm modeling points to heterogeneous multilayer adsorption driven by π–π interactions, hydrogen bonding, and electrostatic forces. In simpler terms, the nanoparticles don’t just trap contaminants; they engage with them on a molecular level.
To optimize performance, the team applied advanced modeling techniques: Response Surface Methodology (RSM) for ibuprofen and Taguchi design for naproxen. Both approaches identified solution pH and adsorbent dosage as the most influential parameters, offering a clear roadmap for scaling up the technology. “This isn’t just academic,” says Mollai Tavani. “We’re talking about a system that can be tuned for real-world conditions—variable pH, fluctuating contaminant loads, and continuous operation.”
Published in *Desalination and Water Treatment* (Desalinare va Pakize Karde Aab), the study underscores the scalability potential of Fe₃O₄–P4VP nanoparticles. Their superparamagnetic property ensures rapid separation, while the polymer’s functional groups can be tailored for other emerging contaminants. For industries grappling with water reuse mandates and tightening environmental regulations, this research offers more than a solution—it presents a paradigm shift.
As energy companies seek to reduce freshwater consumption and minimize wastewater discharge, innovations like these could redefine the balance between operational efficiency and environmental stewardship. The next step? Proving the technology in pilot-scale water treatment systems, where variables like flow rate, fouling, and long-term stability come into play. But for now, the foundation is solid—and the magnetic pull toward cleaner water has never been stronger.
The work, rooted in environmental engineering, may soon find its way from lab benches in Karaj to treatment plants across the globe, proving that sometimes, the most powerful solutions are the ones you can’t see—until you need them.