In a Cairo lab, chemistry professor Aly Reda and his team at The American University in Cairo have turned a dual headache—industrial waste and water pollution—into a breakthrough that could reshape how heavy metals are monitored in energy and manufacturing sectors.
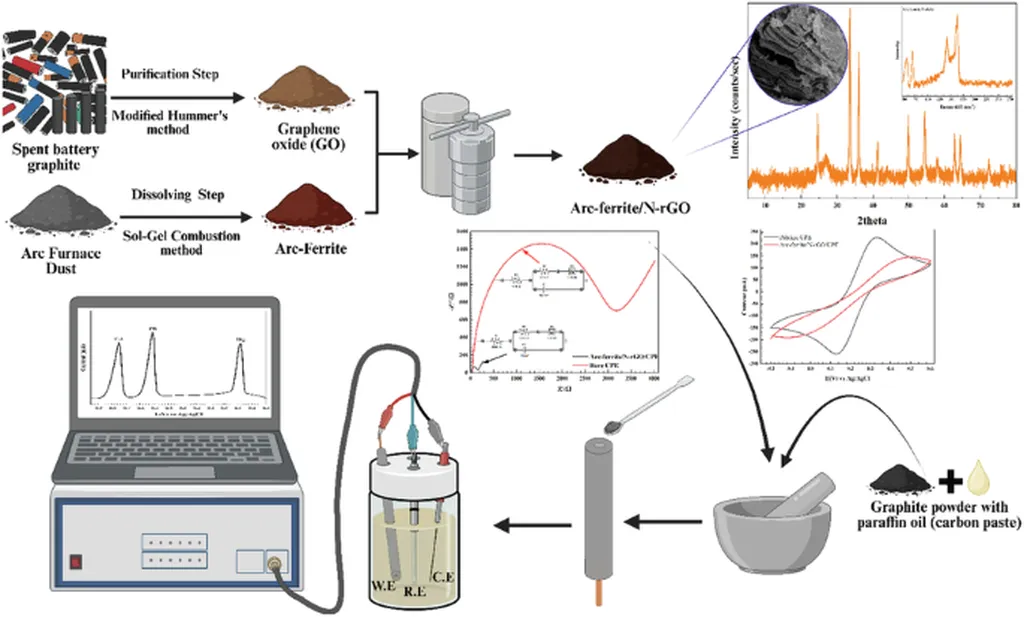
Their innovation, published in *npj Clean Water* (Journal of Clean Water in Arabic), centers on repurposing two common waste streams: arc furnace dust from steelmaking and graphite from spent batteries. By converting these materials into a high-performance nanocomposite sensor, they’ve created a low-cost, highly sensitive tool for detecting toxic heavy metals like lead, cadmium, and mercury in water.
“Instead of seeing waste as a problem, we’re treating it as a resource,” Reda said. “What was once an environmental liability is now a technological asset.”
The core of the advance lies in the Arc-ferrite/N-rGO nanocomposite, where ferrite nanoparticles from steel dust are embedded in nitrogen-doped reduced graphene oxide derived from battery graphite. When integrated into a carbon paste electrode, this composite dramatically enhances electrochemical signals—boosting detection sensitivity by 3.5 times compared to conventional electrodes.
The sensor operates under optimized conditions: a mild acidic buffer at pH 4.5, a deposition potential of -1.1 volts, and a 3-minute pre-concentration step. It can detect lead, cadmium, and mercury at parts-per-billion levels—well below World Health Organization limits for drinking water—with detection limits as low as 1.01 ppb.
What makes this especially compelling for energy and industrial operators is its real-world validation. The team tested the sensor on industrial wastewater, accurately measuring heavy metal concentrations that were cross-checked using advanced spectroscopy. It also showed strong resistance to interference from other common ions, even at 100-fold excess—a critical feature for complex industrial effluents.
For sectors like oil and gas, mining, and thermal power generation—where heavy metal discharge is closely regulated—the implications are significant. Traditional analytical methods require expensive instrumentation and trained personnel, often with long turnaround times. This sensor, by contrast, offers a portable, low-cost alternative that can be deployed on-site, enabling faster, more frequent monitoring and better compliance management.
Reda emphasizes the dual benefit: “We’re not just improving detection—we’re reducing waste and lowering costs at the same time.”
The study, published in *npj Clean Water* (Journal of Clean Water), demonstrates a scalable model for circular economy innovation in environmental sensing. As industries face increasing pressure to minimize both pollution and waste, technologies that convert trash into tools could become a cornerstone of next-generation environmental monitoring.
With detection limits rivaling lab-based instruments and a manufacturing process rooted in waste valorization, this research points toward a future where sustainability and sensing go hand in hand—one where every factory’s byproduct could become a detector of its own pollution.