In the shadow of abandoned mines, where acidic runoff once spelled environmental ruin, a breakthrough from South Africa is turning a costly liability into a valuable asset. Researchers at the University of South Africa (UNISA), led by Mamile Belina Mahlohla from the Centre for Integrated Water Resource Management, have demonstrated how ferric chloride synthesized from iron recovered in acid mine drainage (AMD) can be used to purify drinking water—at less than half the cost of commercial alternatives.
“This isn’t just waste management; it’s waste transformation,” says Mahlohla. “We’re taking what was once a toxic byproduct and turning it into a reagent that meets—and even exceeds—drinking water standards.”
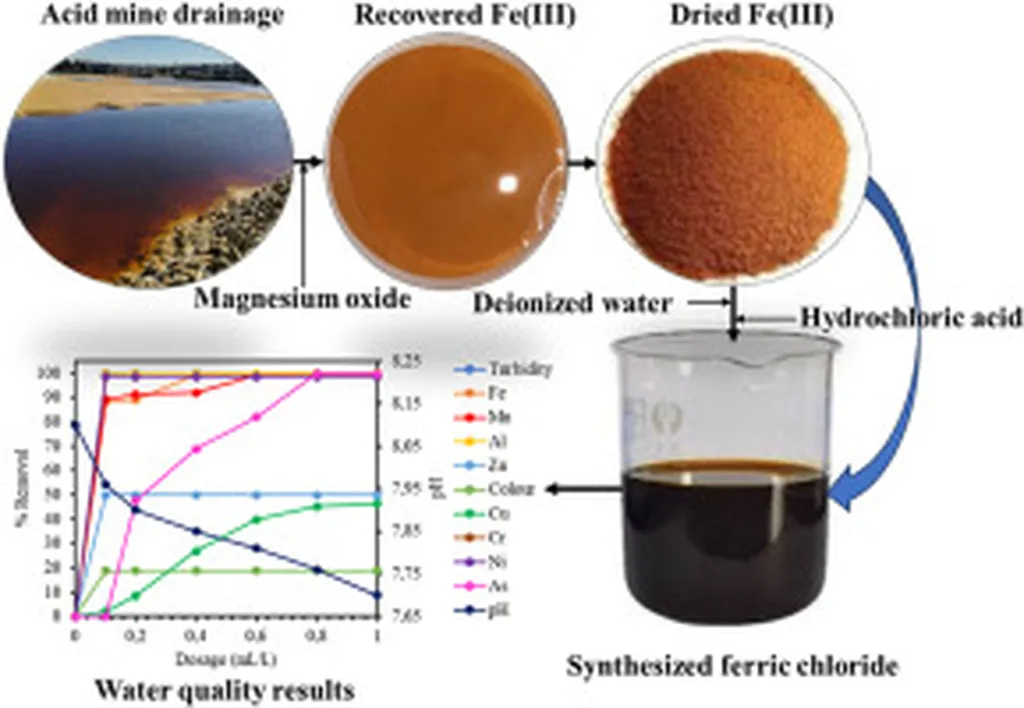
AMD, a persistent challenge in mining regions worldwide, is rich in dissolved iron but also laced with heavy metals and acidity. Traditional neutralization methods often require large volumes of lime or soda ash, generating voluminous sludge and high costs. Mahlohla’s team took a different route: using magnesium oxide nanoparticles derived from local magnesite to selectively precipitate ferric iron (Fe(III)) from AMD. The process avoids the co-precipitation issues seen with calcium- or sodium-based agents, yielding a cleaner iron-rich precipitate.
That precipitate was then dissolved in hydrochloric acid to produce ferric chloride (FeCl₃)—a coagulant widely used in water treatment to remove turbidity, heavy metals, and pathogens. When tested on South African river water, the AMD-synthesized FeCl₃ achieved over 99% removal of turbidity, iron, and aluminum under optimized conditions (0.2 mL/L dose, 100 rpm mixing, 5-minute contact time), fully complying with South African National Standards (SANS 241) for potable water.
What makes this finding significant beyond the lab is its potential to stabilize supply chains for coagulants—a critical input for the energy sector, especially in regions with water-intensive operations like coal or mineral processing. Commercial ferric chloride prices fluctuate due to global supply constraints and energy costs. By tapping into AMD, a waste stream already present at mine sites, water treatment plants—and even power stations—could reduce dependence on imported reagents.
“For industries managing large water streams, like thermal power plants or desalination facilities, this could mean more predictable costs and a lower carbon footprint,” Mahlohla notes.
The research, published in *Water Resources and Industry* (formerly known as *Water Resources and Industry* in Afrikaans: *Waterbronne en Nywerheid*), also hints at broader implications: by partially neutralizing AMD during iron recovery, the door opens for further water reclamation—turning a once-toxic discharge into a source of clean water.
As the global mining and energy sectors face increasing pressure to adopt circular economy principles, this study offers a compelling model: a closed-loop solution where waste from one process becomes the feedstock for another. It’s not just about cleaning water—it’s about reimagining waste as a strategic resource.
And with AMD continuing to threaten water security across South Africa, Australia, the U.S., and beyond, innovations like this could redefine how industries—and entire regions—view their environmental footprints.