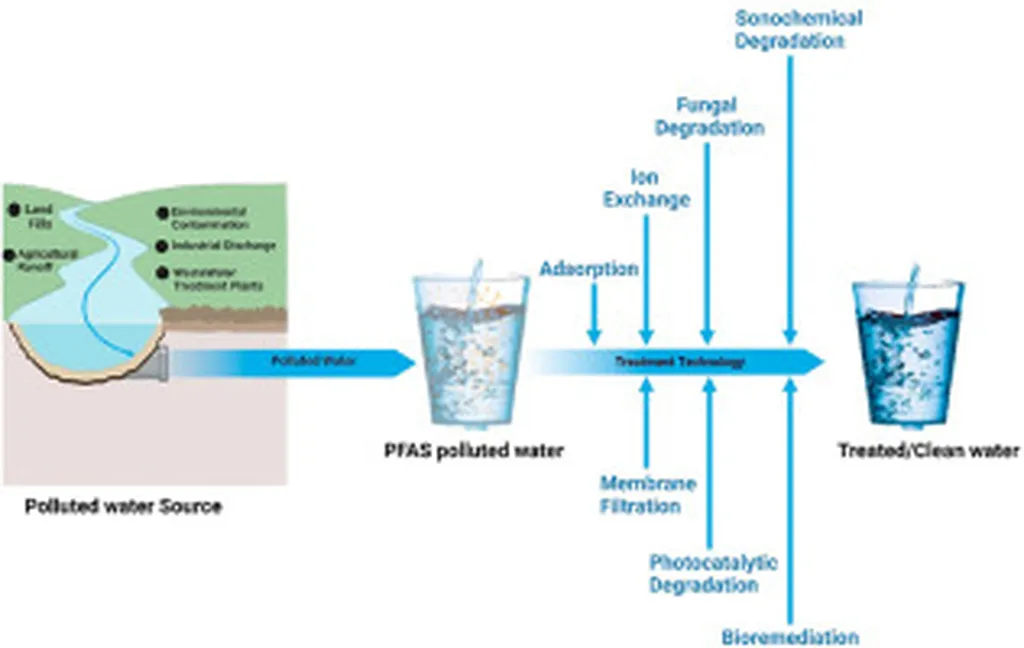
Baiwen Ma’s team at the Chinese Academy of Sciences has turned a spotlight on a quietly escalating crisis: the cocktail of “forever chemicals” and other emerging contaminants that conventional waterworks were never designed to remove. Their review, published in *Fundamental Research*, pulls together the latest evidence and warns that today’s coagulants and sand filters are leaving behind a growing residue of low-molecular-weight compounds that slip through treatment and can re-form into even more toxic by-products when chlorine or ozone is applied. “We are finding that the very processes we rely on to make water safe can, in some cases, open a back door to new hazards,” Ma notes.
For energy-intensive industries—power plants, refineries, data centers, and the sprawling supply chains behind them—the implications are immediate. Cooling towers, boiler feed-water, and high-purity rinse lines all demand water that meets increasingly stringent limits on per- and polyfluoroalkyl substances (PFAS), pharmaceuticals, microplastics, and endocrine disruptors. Where a decade ago utilities could offset inadequate removal with deeper beds of granular activated carbon or higher doses of disinfectant, those fixes now carry their own regulatory and energy penalties. “Each additional adsorption stage or oxidation step adds megawatt-hours of pumping, heating, or UV lamp load,” explains Ma. “If we can’t target the contaminants at source, the energy penalty becomes structural.”
The paper maps four technology families—coagulation, adsorption, membrane filtration, and advanced oxidation—and shows how hybrid “treatment trains” can cut the energy intensity of removal by up to 40 % when the right sequence is matched to contaminant chemistry. For example, a weak anion exchange resin placed upstream of ultrafiltration can capture PFAS before they foul the membrane, reducing the frequency of expensive chemical cleaning cycles. Another example is the pairing of electro-oxidation with ceramic microfiltration; the oxidation step mineralizes the contaminants while the membrane retains the catalyst, cutting reagent use by more than half compared with conventional ozonation.
Energy operators are already piloting some of these combinations. A 500 MW combined-cycle plant in Jiangsu province has retrofitted a side-stream PFAS polishing loop using ion-exchange resin regenerated on-site with brine, slashing both resin replacement costs and truck rolls. The data suggest that retrofits can pay back in 18–24 months when compared with the alternative of deepening GAC beds or building new nanofiltration units.
Ma’s team also flags the next wave of materials: metal-organic frameworks tailored for specific functional groups, boron-doped diamond electrodes that resist fouling, and biochar derived from agricultural waste that can be regenerated with low-temperature steam. “The materials science is moving faster than the standards,” observes Ma. “Utilities and industrial water managers need to start running parallel pilot tests now, because the approval cycle for a new adsorbent or membrane can take three to five years—longer than the regulatory clock is ticking.”
What emerges from the review is less a single breakthrough and more a toolkit for risk-weighted design. Energy companies can use the contaminant-property matrix provided by Ma’s group to rank their sites by both exposure risk and energy intensity, then deploy the least-energy-intensive sequence that still meets future discharge limits. The paper itself is a call to action for materials chemists, process engineers, and plant operators to co-develop solutions that are not just technically effective but also energy-aware. Published in *Fundamental Research*, the work offers a scientific foundation that the industry can build on—before the next round of drinking-water standards forces the issue.