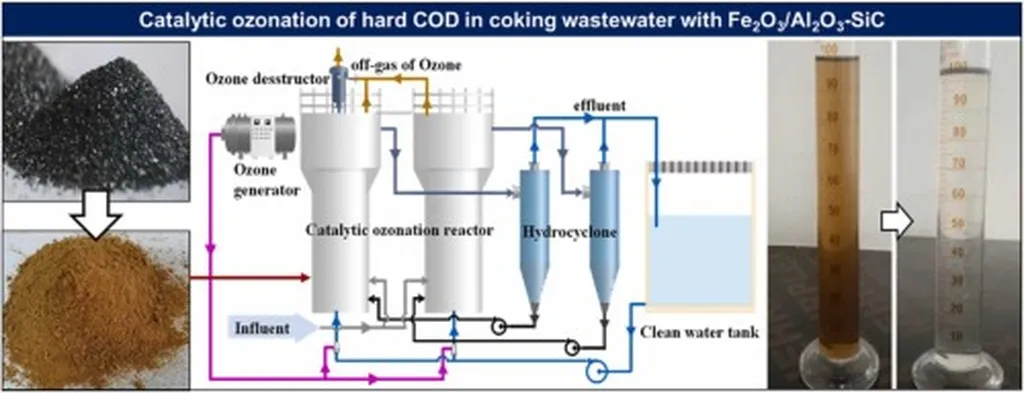
The coking industry faces a persistent challenge: how to treat its wastewater to meet increasingly stringent environmental standards without breaking the bank. Beijing-based researcher Wang Guanying, from Beijing Boqi Electric Power Science and Technology Co., Ltd., has taken a significant step toward solving this puzzle. In a new study published in *Gongye shui chuli* (Industrial Water Treatment), Wang and colleagues explore how ozone catalytic oxidation could revolutionize the advanced treatment of coking wastewater—offering both environmental compliance and economic viability for power and steel plants.
Coking wastewater is notoriously difficult to treat. It contains high concentrations of toxic organic compounds, ammonia, cyanide, and phenols—residues from the high-temperature processing of coal into coke. Traditional biological treatment can remove much of the organic load, but it often leaves behind persistent pollutants that require advanced oxidation processes (AOPs) to break down further. Ozone oxidation has emerged as a leading candidate for this final polishing step, but it comes with two major hurdles: high energy costs for ozone generation and low utilization efficiency of the ozone itself.
Wang’s team conducted a comprehensive review of existing research and found that while ozone oxidation is widely used, its efficiency can be dramatically improved through catalytic enhancement. By comparing four catalytic approaches—adsorption materials, micro/nano bubbles, peroxide oxidation, and active metal catalysts—they concluded that active metal catalytic ozone oxidation stands out as the most promising for coking wastewater.
“Active metal catalytic ozone oxidation not only improves the degradation of refractory organics but also significantly reduces ozone demand,” Wang noted. In their analysis, the addition of metal catalysts increased Total Organic Carbon (TOC) and Chemical Oxygen Demand (COD) removal by 12% to 42% compared to ozone alone. That’s a game-changer for plant operators looking to cut both costs and carbon footprints.
The study also provides practical guidance for implementation. Optimal conditions for the process include a temperature of 20–25°C, a neutral or weakly alkaline pH, and an ozone-to-COD ratio of 0.25–0.70 mg/mg. Adjusting the wastewater pH to near the catalyst’s zero-charge point can further boost efficiency, as it enhances the generation of hydroxyl radicals (·OH), the powerful oxidants responsible for breaking down pollutants.
But the research doesn’t stop at performance metrics—it dives into the chemical intricacies. Common ions like chloride, sulfate, and carbonate—abundant in coking wastewater—can inhibit catalytic activity. Wang’s team suggests that understanding how these Lewis bases interact with catalysts could guide the design of more resilient materials, potentially turning a problem into a solution.
For energy-intensive industries like steel and power generation, where coking is a key process, this research offers a tangible pathway to cleaner operations. By integrating active metal catalytic ozone oxidation into wastewater treatment systems, plants could not only meet zero-discharge targets but also reduce chemical and energy consumption. That translates to lower operational costs and a smaller environmental footprint—critical factors in an era of tightening regulations and rising scrutiny over industrial emissions.
As the coking industry continues to evolve under pressure to decarbonize and comply with environmental laws, innovations like those highlighted in *Industrial Water Treatment* will be pivotal. Wang’s work underscores a growing trend: the fusion of advanced chemistry with industrial pragmatism. It’s not just about treating wastewater—it’s about redefining what’s possible in sustainable industrial operations.