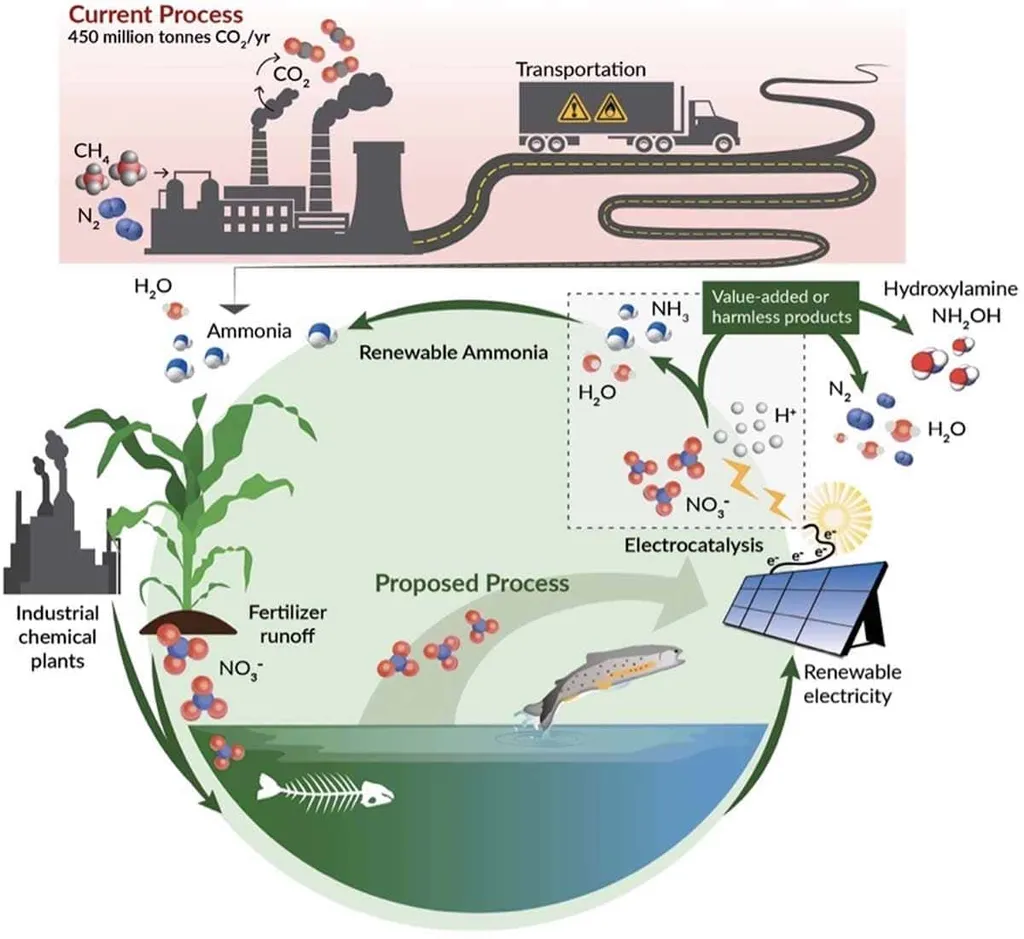
The race to clean up our waterways while extracting value from pollution has taken a promising turn, thanks to a breakthrough synthesis of electrochemistry and materials science. Researchers at Wuhan University, led by Professor Huiling Wang, have published a comprehensive review in *Environmental Chemistry and Safety* (环境化学与安全) that reimagines how we handle nitrate-laden wastewater—not just as a problem to remove, but as a potential resource to recover.
Nitrate pollution isn’t just an environmental issue; it’s a growing industrial headache. Runoff from fertilizers, industrial discharges, and aging infrastructure has elevated nitrate levels in rivers and aquifers worldwide, threatening ecosystems and public health. Traditional remediation methods like reverse osmosis or biological denitrification are effective but often costly and energy-intensive, with limited secondary benefits. Enter electrocatalytic nitrate reduction (NO₃RR)—a process that uses electricity to convert harmful nitrates into useful compounds like ammonia, a cornerstone of fertilizer production.
What makes this approach especially compelling is its alignment with the energy transition. “Electrocatalysis allows us to couple wastewater treatment with renewable energy sources,” explains Professor Wang. “Instead of treating nitrate removal as a standalone cost center, we’re turning it into a node in a circular economy—where pollution becomes feedstock and electrons become reagents.” This dual-purpose model could reduce the carbon footprint of both water treatment and chemical manufacturing.
The science behind the transformation is intricate. NO₃RR doesn’t follow a straight path. Nitrate (NO₃⁻) gets reduced through a series of intermediates—nitrite (NO₂⁻), nitric oxide (*NO), and nitrous oxide—before forming ammonia (NH₃) or even coupling with carbon to produce urea or amino acids. The efficiency and product outcome depend heavily on the catalyst. Recent advances highlighted in the review include single-atom catalysts, alloy nanostructures, and materials engineered with atomic vacancies—each designed to stabilize key intermediates and steer the reaction toward the desired product.
One of the most exciting developments is electrochemical C–N coupling, where nitrate and organic carbon sources react under mild conditions to form high-value chemicals like urea or oximes. “This isn’t just about cleaning water,” says Wang. “It’s about using the energy from renewables to build molecules that would otherwise require high-temperature, high-pressure industrial processes.” Imagine a future where a wastewater treatment plant not only cleans water but also co-produces fertilizer or pharmaceutical intermediates—all powered by solar or wind energy.
Yet challenges remain. Real wastewater contains impurities, competing ions, and fluctuating nitrate concentrations. Catalysts must maintain performance under these dynamic conditions, and reactor designs need to scale efficiently for industrial use. The review emphasizes the need for advanced in situ characterization tools—like synchrotron-based spectroscopies—to watch catalysts evolve in real time and adjust their structure accordingly.
For the energy sector, this research signals a convergence of interests. Utilities and industrial operators are increasingly under pressure to reduce emissions and improve resource efficiency. Electrocatalytic systems could be deployed at the nexus of water and energy infrastructure—treating effluents from power plants, chemical factories, or agricultural runoff while generating products with market value.
As the global push for sustainability intensifies, innovations like NO₃RR offer a glimpse of a future where pollution isn’t just managed, but monetized. The work led by Professor Wang and her team at Wuhan University isn’t just a scientific milestone—it’s a strategic pivot toward systems that deliver environmental, economic, and energy benefits in one integrated process. Published in *Environmental Chemistry and Safety*, the review doesn’t just summarize the state of the art; it maps a pathway forward for industries ready to rethink waste as opportunity.